In instances where bond angles can’t be determined through electronic geometry or VSEPR theory, mathematical equations can provide the solution. The following are molecular geometries with their corresponding bond angles obtained from VSEPR theory: Determine the bond angles using the molecular geometry. Use VSEPR theory to predict the molecular geometry.Į. Determine the number of bonding electron pairs and lone electron pairs on the central atom.ĭ. Determine the molecule’s Lewis structure.Ĭ. By applying VSEPR theory, you can determine the molecule’s shape and calculate bond angles based on its molecular geometry.Ī. VSEPR theory states that regions containing electron pairs (bonding and non-bonding) around a central atom will repel each other and arrange themselves to minimize this repulsion. Valence Shell Electron Pair Repulsion (VSEPR) Theory 
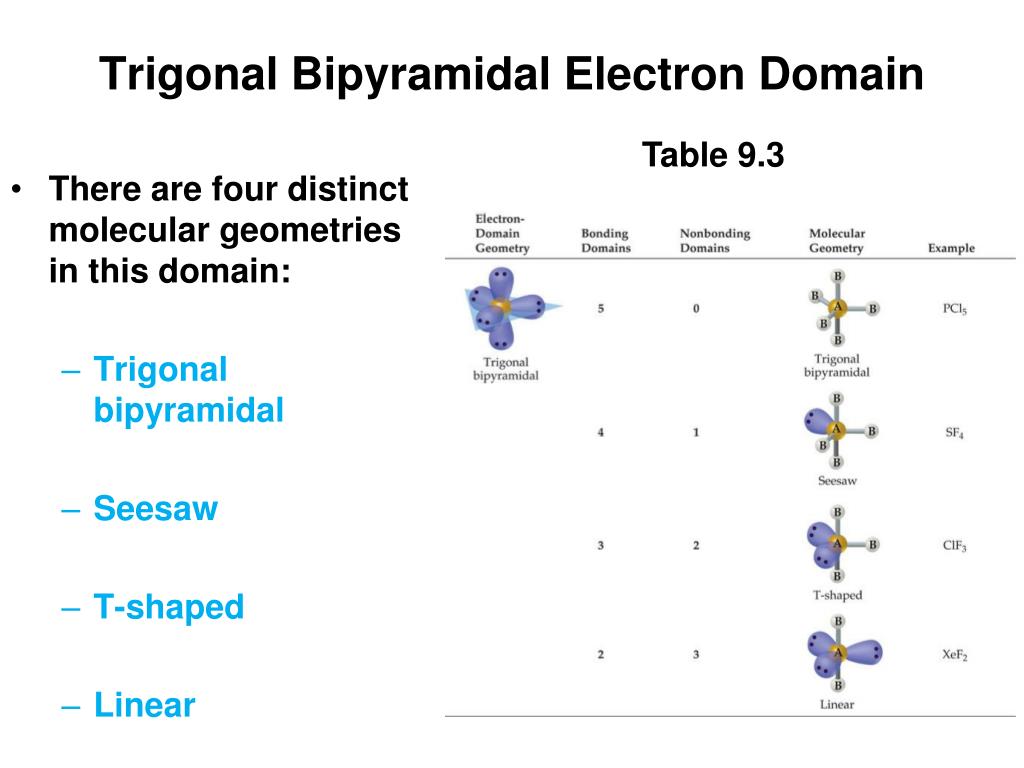
Knowing these primary geometries will help you identify the approximate bond angles in various molecules.Ģ. These geometries exhibit specific bond angles: The five primary electronic geometries are linear, trigonal planar, tetrahedral, trigonal bipyramidal, and octahedral. Each electron groups’ position affects the bond angles in a molecule. 
This article will guide you on how to determine bond angles using three different methods: electronic geometry, valence shell electron pair repulsion (VSEPR) theory, and mathematical equations.Įlectronic geometry, also known as the parent shape or domain shape, provides an overall picture of the electron distribution around the central atom in a molecule. Understanding how to calculate bond angles is crucial for anyone studying chemistry, as it enables them to comprehend molecular geometry and predict a compound’s behavior. A bond angle is defined as the angle formed between two covalent bonds that connect one atom to another, creating a geometric arrangement of atoms within a molecule. Bond angles play a significant role in defining the shape and properties of molecules. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
